Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Honda, Mitsunori; Shimoyama, Iwao; Okamoto, Yoshihiro; Suzuki, Shinichi; Yaita, Tsuyoshi
no journal, ,
no abstracts in English
Oikawa, Kenichi; Harada, Masahide; Oi, Motoki; Kai, Tetsuya; Shinohara, Takenao; Sakai, Kenji; Maekawa, Fujio
no journal, ,
The day-1 instrument NOBORU at BL10 in MLF/J-PARC was constructed by the neutron source development group of MLF. We have carried out general use and project use program since 2008. The beam-line component of the instrument was upgraded step by step, and evaluated by the instrument group. The PLC control system was also adopted in this winter. We will present performance evaluation of these components and introduce the new control system for these apparatus.
Baba, Yuji; Shimoyama, Iwao; Hirao, Norie; Sekiguchi, Tetsuhiro
no journal, ,
no abstracts in English
Ito, Takashi
no journal, ,
no abstracts in English

Hirano, Yu; Kimura, Shigenobu*; Tamada, Taro
no journal, ,
Cytochrome  (Cyt
(Cyt  ) consisting of approximately 134 amino acid residues is a heme-binding protein and binds to the endoplasmic reticulum (ER) membrane. The N-terminal region of Cyt
) consisting of approximately 134 amino acid residues is a heme-binding protein and binds to the endoplasmic reticulum (ER) membrane. The N-terminal region of Cyt  is the heme-binding domain in the cytoplasmic side of ER membrane and the C-terminal region is the membrane-binding domain. We performed X-ray crystal structure analyses of the N-terminal 94 residues of the Cyt
is the heme-binding domain in the cytoplasmic side of ER membrane and the C-terminal region is the membrane-binding domain. We performed X-ray crystal structure analyses of the N-terminal 94 residues of the Cyt  from porcine liver in both oxidized and reduced states. Diffraction experiments were carried out at the BL17A beamline of PF. We have obtained the data sets at 0.85
from porcine liver in both oxidized and reduced states. Diffraction experiments were carried out at the BL17A beamline of PF. We have obtained the data sets at 0.85  , resolution (+Ca) and 0.93
, resolution (+Ca) and 0.93  , resolution (-Ca) for the oxidized form and at 0.85
, resolution (-Ca) for the oxidized form and at 0.85  , resolution (+Ca) and 0.98
, resolution (+Ca) and 0.98  , resolution (-Ca) for the reduced form. Structure refinement revealed that the side chains of two glutamic acid residues near the heme group were involved in the coordination of a Ca ion in the crystal structure with Ca ions.
, resolution (-Ca) for the reduced form. Structure refinement revealed that the side chains of two glutamic acid residues near the heme group were involved in the coordination of a Ca ion in the crystal structure with Ca ions.
Tamada, Taro; Arimori, Takao*; Fukuhara, Hiroaki*; Ito, Akihiro*; Ueda, Mitsuhiro*
no journal, ,
The saccharification process is essential for bioethanol production from woody biomass including celluloses. Cold-adapted cellulase, which has sufficient activity at low temperature ( 293 K), is capable of reducing heating costs during the saccharification process and is suitable for simultaneous saccharification and fermentation. Endo-1,4-
293 K), is capable of reducing heating costs during the saccharification process and is suitable for simultaneous saccharification and fermentation. Endo-1,4- -glucanase from the earthworm Eisenia fetida (EF-EG2) belonging to glycoside hydrolase family 9 has been shown to have the highest activity at 313 K, and also retained a comparatively high activity at 283 K. The recombinant EF-EG2, expressed in
-glucanase from the earthworm Eisenia fetida (EF-EG2) belonging to glycoside hydrolase family 9 has been shown to have the highest activity at 313 K, and also retained a comparatively high activity at 283 K. The recombinant EF-EG2, expressed in 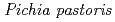 , was purified and then grew needle-shaped crystals with dimensions of 0.02
, was purified and then grew needle-shaped crystals with dimensions of 0.02  0.02
0.02  1 mm. The final model of EF-EG2, including 435 residues, two ions, seven crystallization reagents and 696 waters, was refined to a crystallographic
1 mm. The final model of EF-EG2, including 435 residues, two ions, seven crystallization reagents and 696 waters, was refined to a crystallographic 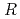 -factor of 14.7% (free
-factor of 14.7% (free 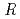 -factor of 16.8%) to 1.5
-factor of 16.8%) to 1.5  resolution. The overall structure of EF-EG2 has an(
resolution. The overall structure of EF-EG2 has an( /
/ )
) barrel fold which contains a putative active-site cleft and a negatively charged surface. Furthermore, in the structure of the E431Q mutant of EF-EG2 complexed with cellotriose (CTR), electron densities corresponding to CTR are observed well at -4 to -2 subsites. This structural information helps us understand the catalytic and cold adaptation mechanisms of EF-EG2.
barrel fold which contains a putative active-site cleft and a negatively charged surface. Furthermore, in the structure of the E431Q mutant of EF-EG2 complexed with cellotriose (CTR), electron densities corresponding to CTR are observed well at -4 to -2 subsites. This structural information helps us understand the catalytic and cold adaptation mechanisms of EF-EG2.
 O
O by using PDF analysis
by using PDF analysisKodama, Katsuaki; Igawa, Naoki; Shamoto, Shinichi; Ikeda, Kazutaka*; Oshita, Hidetoshi*; Kaneko, Naokatsu*; Otomo, Toshiya*; Suzuya, Kentaro; Hoshikawa, Akinori*; Ishigaki, Toru*
no journal, ,
no abstracts in English
Kuroki, Ryota
no journal, ,
no abstracts in English
 O
O observed by PDF analysis
observed by PDF analysisKodama, Katsuaki; Igawa, Naoki; Shamoto, Shinichi; Ikeda, Kazutaka*; Oshita, Hidetoshi*; Kaneko, Naokatsu*; Otomo, Toshiya*; Suzuya, Kentaro; Hoshikawa, Akinori*; Ishigaki, Toru*
no journal, ,
no abstracts in English
Aso, Tomokazu; Tatsumoto, Hideki; Otsu, Kiichi; Kawakami, Yoshihiko; Komori, Shinji; Muto, Hideki; Takada, Hiroshi
no journal, ,
no abstracts in English
Kanari, Yukiko; Noguchi, Miho; Kaminaga, Kiichi; Sakamoto, Yuka; Yokoya, Akinari; Suzuki, Keiji*
no journal, ,
no abstracts in English
Kaminaga, Kiichi; Narita, Ayumi; Noguchi, Miho; Kanari, Yukiko; Sakamoto, Yuka; Yokoya, Akinari
no journal, ,
no abstracts in English
Sakamoto, Yuka; Kanari, Yukiko; Kaminaga, Kiichi; Noguchi, Miho; Yokoya, Akinari
no journal, ,
no abstracts in English
Kajimoto, Ryoichi; Nakamura, Mitsutaka; Inamura, Yasuhiro; Kamazawa, Kazuya*; Ikeuchi, Kazuhiko*; Iida, Kazuki*; Ishikado, Motoyuki*; Suzuki, Junichi*; Nakajima, Kenji; Kawamura, Seiko; et al.
no journal, ,
no abstracts in English
Fukaya, Yuki; Mochizuki, Izumi*; Maekawa, Masaki; Wada, Ken*; Hyodo, Toshio*; Matsuda, Iwao*; Kawasuso, Atsuo
no journal, ,
no abstracts in English
Narita, Ayumi; Kaminaga, Kiichi; Yokoya, Akinari; Noguchi, Miho; Kobayashi, Katsumi*; Usami, Noriko*; Fujii, Kentaro
no journal, ,
no abstracts in English
Narita, Ayumi; Fujii, Kentaro; Yokoya, Akinari; Baba, Yuji; Shimoyama, Iwao
no journal, ,
no abstracts in English